The co-infection of HIV-1 viruses has emerged as a significant threat to global public health as a result of shared mode of transmission. This article presents a novel mathematical model that addresses the dynamics of this co-infection by extending the SVEIR (Susceptible – Vaccinated – Exposed – Infectious - Recovered) framework to incorporate time-delay, chemotherapy and quarantine compartments. The population is divided into twelve compartments, with infections individuals further subdivided into symptomatic and asymptomatic individuals. The mathematical model developed is constrained to adhere to fundamental epidemiology properties such as non-negativity and boundedness within a feasible. We investigate the fundamental reproduction number that guarantees stability of equilibrium points are disease free and endemic qualitative behavior of models are examined. Stability threshold explicitly state that when reproduction number is less than one the disease free equilibrium is globally asymptotically stable, meaning the infection can be eliminated. Using Lyapunov functions, local and global stability of these states are explored and findings presented graphically. They were used to account for the history dependent nature of time delay. Our research assessed control policies and proposed alternatives, performing bifurcation analysis so as to establish prevention strategies. We investigated Hopf bifurcation analytically and numerically to demonstrate disease dynamics, which is novel to our study.. Numerical simulations, performed using the MATLAB dde23 solver, demonstrate that the introduction of chemotherapy and quarantine significantly reduces the peak of symptomatic infections. Crucially, our Hopf bifurcation analysis identifies a critical delay threshold beyond which stable equilibrium is lost to sustained periodic oscillations, representing recurrent waves of infection or rather viral blips. This offered new insights into the long-term management of HIV-1 co-infection cycles.
| Published in | Mathematical Modelling and Applications (Volume 11, Issue 1) |
| DOI | 10.11648/j.mma.20261101.12 |
| Page(s) | 18-27 |
| Creative Commons |
This is an Open Access article, distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium or format, provided the original work is properly cited. |
| Copyright |
Copyright © The Author(s), 2026. Published by Science Publishing Group |
Coronavirus, Basic Reproduction Number, Global Stability, Lyapunov’s Function, Bifurcation

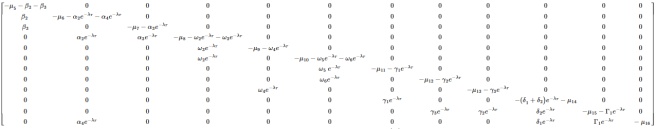 (13)
(13) Parameters | Value | Source |
|---|---|---|
| 2500 | Fixed |
| 1000 | Estimated |
| 1500 | Estimated |
| 100 | Estimated |
| 10 | Estimated |
| 10 | Estimated |
| 0 | Assumed |
| 0 | Assumed |
| 0 | Assumed |
| 15 | Estimated |
| 20 | Estimated |
| 5 | Assumed |
| 2500 | 16 |
| 0.20 | Assumed |
| 0.002 | 14 |
| 0.53 | Estimated |
| 0.45 | Estimated |
| 0.50 | Estimated |
| 0.4 | Estimated |
| 0.05 | Estimated |
| 0.043 | Estimated |
| 0.045 | Estimated |
| 0.3425 | Estimated |
| 0.05 | 22 |
| 0.3 | 16 |
| 0.3 | 16 |
| 0.38 | 16 |
| 0.200 | 22 |
| 0.3 | Assumed |
|
| 16 |
DDE | Delay Differential Equations |
DFE | Disease Free Equilibrium |
EEP | Endemic Equilibrium Point |
HIV | Human Immuno Deficiency Virus |
J | Jacobian |
WHO | World Health Organisation |
| [1] | Byul Nim, Eunjung Kim, Sunmi Lee, and Chunyoung Oh, 2020, Mathematical Model of COVID-19 Transmission Dynamics in South Korea The Impact of Tavel Restrictions, Social Distancing and Early Detection, Journal of Maths MDPI Publishers pg 1-18. |
| [2] | Sarbaz H. A. Khoshnaw, Rizgar H. Salih and Sadegh Sulaimany, 2020, Mathematical Modelling for Coronavirus diseases (COVID-19) in Predicting future behaviours and sensitivity Analysis, Journal of Mathematical Modelling of Natural Phenomena. |
| [3] | Anwar Zeb, Ebraheem Alzahrani, Vedat Suat Erturk and Gul Zaman, 2020, Journal of Biomed Research International Hindawi Publishers, volume 2020, 7 pages. |
| [4] | Viona Ojiambo, Mark Kimathi, Samuel Mwalili, Duncan Gathungu, Rachel W. Mbogo, 2020, A Human-pathogen SEIR-P Model for COVID-19 Outbreak Under different intervention Scenario in Kenya, journal of Mathematics, pg 1-10. |
| [5] | Pakwan Riyapan, Sherif Eneye Shualb and Arthit Intarasit. 2021. A Mathematical Model of COVID-19 Pandemic A case Study of Bangkok, Thailand, Journal of Computational and Mathematical methods in medicine, Hindawi Publishers, 11 pages. |
| [6] | Betti, M.; Bragazzi N.; Heffernan, J.; Kong, J.; Raad, A. Could a New COVID-19 Mutant Strain Undermine Vaccination Efforts? A Mathematical modelling approach for estimating the spread of B.1.1.7 Using Ontario, Canada as a Case Study Vaccines, 2021, a, 592. |
| [7] | Ali Alarjani, Md Taufiq Nasseef, Sanaa M. Sharif B. V. Subba Rao, Mufti Mahmud and Md Sharif Uddin, 2020, Application of Mathematical modelling in prediction of COVID-19 transmission dynamics, Journal of computer engineering and computer science, Springer open publishers. |
| [8] | Rahim Ud Din, Kamal Shah, Imtiaz Ahmad and Thabet Abdelijawed, 2020, study of transmission dynamics of novel COVID-19 by using mathematical model, Journal of Advances in Difference Equations, Springer open publishers, pg 1-13. |
| [9] | Liu, T.; Kang, L.; Li, Y.; Huang, J.; GUO, Z; Xu, J.; Hu, Y.; Zhai, Z.; Kang, X.; Jiang, T.; et al. Simultaneous Detection of seven Human Coronaviruses by multiplex PCR and MALDI-TOFMS. COVID 2022, 2, 5-17. |
| [10] | Elrashdy, F.; E. M.; Uversky, V. N. on the safety of covid-19 convalescent plasma treatment: Thrombotic and Thromboembolic concerns. COVID 2022, 2, 1-4. |
| [11] | Hirota, K.; Mayahara, T.; Fujii, Y.; Nishi, K. Asymptomatic Hypoxemia as a characteristic symptom of coronavirus Disease. A narrative Review of its pathophysiology. COVID 2022, 2, 47-59. |
| [12] | Chable-Bessia, C,; Boulle, C; Neyret, A.; Swain, J.; Henaut, M.; Merida, P.; Gros, N.; Makinson, A.; Lyonnais, S.; Chesnais, C.; et al. Low selectivity indices of invermectin and macrocyclic lactones on SARS-COV-2 Replication in vitro. COVID 2022, 60-75. |
| [13] | Kaur, A.; Michalopous C.; Carpe, S; Congrete, S.; Shahzad, H,; Reardon, J.; Wakefield, D.; Swart, C.; Zuwallack, R. Post- covid-19 condition and health status 2022, 2, 76-86 (MDPI) Publishers. |
| [14] | Peronace, C.; Tallerico, R.; Colosimo, M.; Panduri, G.; Pintomalli, L.; Oteri R.; Calantoni, V.; et al (2022) B A. I Omicron Variant of SARS-COV-2: First Case Reported in Calabria Region, Italy. COVID 2022, 2, 211-215. |
| [15] | Schlickeiser, R.; Kroger, M. Forecast of Omicron Wave Time Evolution. COVID 2022, 2, 216-229. |
| [16] | Cho, D-H.; Choi, J.; Gwon, J. G. Atorvastatin Reduces Severity of COVID-19: A Nationwide, Total population-Based, case-control study. COVID 2022, 2, 398-406. |
| [17] | Sorensen, C. A.; Clemmensen, A.; Sparrewath, C.; Tetens, M. M.; Krogfelt, K. A. Children Naturally Evading COVID-19-Why Children Differ from Adults. COVID 2022, 2, 369-378. |
| [18] | Batra, A.; Swaby, J.; Raval, P.; Zhu, H.; Weintraub, N. L.; Terris, M.; Karim, N. A.; Keruakous, A.; Gulterman, D.; Beyer, K.; et al. Effect of community and socio-economic factors on cardiovascular, cancer and cardio-oncology patients with COVID-19. COVID 2022, 2, 350-368. |
| [19] | Mbhiza and Muthelo (2022) COVID-19 and the Quality of mathematics education, teaching and learning in a first-year course. South African Journal of Higher education volume 36 Number 2 pages 189-203. |
| [20] | N. Ringa, M. L. Diagne, H. Rwezaura, A. Omame, S. Y. Tchoumi and J. M. Tchuenche (2022) HIV and COVID-19 co-infection: A mathematical model and optimal control. Journal of informatics medicine Elsevier publishers page 1-17 |
| [21] | Kassahun Getnet Mekonen, Shiferaw Feyissa Balcha, Legesse Lemecha Obsu and Abdulkadir Hassen (2022) Mathematical modeling and Analysis of TB and COVID-19 Coinfection. Journal of applied mathematics Hindawi publishers pages 1-20. |
| [22] | Kotola, B. S., Teklu, S. W., and Abebaw, Y. F. (2023). Bifurcation and optimal control analysis. |
| [23] | Kirui W., R. K. (2015). Modelling the effects of time delay on HIV-1 in vivo dynamics in the presence of ARVs. Science Journal of Applied Mathematics and Statistics, 204-213. |
| [24] | O. Diekmann, J. H. (1990). On the definition and computation of Ro in models for infectious diseases in heterogeneous populations. Journal of Mathematical Biology, 365-382. |
| [25] | Watmough, P. D. (2002). Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Journal of Mathematical Biosciences, 29-48. |
| [26] | Lawrence Shampine and Skip Thompson (2000), Solving delay differential equations with dde23, Southern Methodist University and Radford University, pages 1-44. |
| [27] | Hezam Ibrahim M., F. A. (2021). A dynamic optimal control model for COVID-19 and cholera co-infection in Yemen. Advances in Difference Equations. |
APA Style
Pela, C., Wesley, K., Daniel, A. (2026). Bifurcation and Stability Analysis of HIV-1 Coronavirus Co-infection Model. Mathematical Modelling and Applications, 11(1), 18-27. https://doi.org/10.11648/j.mma.20261101.12
ACS Style
Pela, C.; Wesley, K.; Daniel, A. Bifurcation and Stability Analysis of HIV-1 Coronavirus Co-infection Model. Math. Model. Appl. 2026, 11(1), 18-27. doi: 10.11648/j.mma.20261101.12
@article{10.11648/j.mma.20261101.12,
author = {Cherono Pela and Kirui Wesley and Adicka Daniel},
title = {Bifurcation and Stability Analysis of HIV-1 Coronavirus
Co-infection Model},
journal = {Mathematical Modelling and Applications},
volume = {11},
number = {1},
pages = {18-27},
doi = {10.11648/j.mma.20261101.12},
url = {https://doi.org/10.11648/j.mma.20261101.12},
eprint = {https://article.sciencepublishinggroup.com/pdf/10.11648.j.mma.20261101.12},
abstract = {The co-infection of HIV-1 viruses has emerged as a significant threat to global public health as a result of shared mode of transmission. This article presents a novel mathematical model that addresses the dynamics of this co-infection by extending the SVEIR (Susceptible – Vaccinated – Exposed – Infectious - Recovered) framework to incorporate time-delay, chemotherapy and quarantine compartments. The population is divided into twelve compartments, with infections individuals further subdivided into symptomatic and asymptomatic individuals. The mathematical model developed is constrained to adhere to fundamental epidemiology properties such as non-negativity and boundedness within a feasible. We investigate the fundamental reproduction number that guarantees stability of equilibrium points are disease free and endemic qualitative behavior of models are examined. Stability threshold explicitly state that when reproduction number is less than one the disease free equilibrium is globally asymptotically stable, meaning the infection can be eliminated. Using Lyapunov functions, local and global stability of these states are explored and findings presented graphically. They were used to account for the history dependent nature of time delay. Our research assessed control policies and proposed alternatives, performing bifurcation analysis so as to establish prevention strategies. We investigated Hopf bifurcation analytically and numerically to demonstrate disease dynamics, which is novel to our study.. Numerical simulations, performed using the MATLAB dde23 solver, demonstrate that the introduction of chemotherapy and quarantine significantly reduces the peak of symptomatic infections. Crucially, our Hopf bifurcation analysis identifies a critical delay threshold beyond which stable equilibrium is lost to sustained periodic oscillations, representing recurrent waves of infection or rather viral blips. This offered new insights into the long-term management of HIV-1 co-infection cycles.},
year = {2026}
}
TY - JOUR T1 - Bifurcation and Stability Analysis of HIV-1 Coronavirus Co-infection Model AU - Cherono Pela AU - Kirui Wesley AU - Adicka Daniel Y1 - 2026/03/23 PY - 2026 N1 - https://doi.org/10.11648/j.mma.20261101.12 DO - 10.11648/j.mma.20261101.12 T2 - Mathematical Modelling and Applications JF - Mathematical Modelling and Applications JO - Mathematical Modelling and Applications SP - 18 EP - 27 PB - Science Publishing Group SN - 2575-1794 UR - https://doi.org/10.11648/j.mma.20261101.12 AB - The co-infection of HIV-1 viruses has emerged as a significant threat to global public health as a result of shared mode of transmission. This article presents a novel mathematical model that addresses the dynamics of this co-infection by extending the SVEIR (Susceptible – Vaccinated – Exposed – Infectious - Recovered) framework to incorporate time-delay, chemotherapy and quarantine compartments. The population is divided into twelve compartments, with infections individuals further subdivided into symptomatic and asymptomatic individuals. The mathematical model developed is constrained to adhere to fundamental epidemiology properties such as non-negativity and boundedness within a feasible. We investigate the fundamental reproduction number that guarantees stability of equilibrium points are disease free and endemic qualitative behavior of models are examined. Stability threshold explicitly state that when reproduction number is less than one the disease free equilibrium is globally asymptotically stable, meaning the infection can be eliminated. Using Lyapunov functions, local and global stability of these states are explored and findings presented graphically. They were used to account for the history dependent nature of time delay. Our research assessed control policies and proposed alternatives, performing bifurcation analysis so as to establish prevention strategies. We investigated Hopf bifurcation analytically and numerically to demonstrate disease dynamics, which is novel to our study.. Numerical simulations, performed using the MATLAB dde23 solver, demonstrate that the introduction of chemotherapy and quarantine significantly reduces the peak of symptomatic infections. Crucially, our Hopf bifurcation analysis identifies a critical delay threshold beyond which stable equilibrium is lost to sustained periodic oscillations, representing recurrent waves of infection or rather viral blips. This offered new insights into the long-term management of HIV-1 co-infection cycles. VL - 11 IS - 1 ER -