This study offers a comprehensive mathematical and computational investigation of the combined effects of temperature gradient and high sickle cell concentration on blood circulation through a porous atherosclerotic channel in the presence of an applied magnetic field. The model incorporates key physiological and physical mechanisms, including magnetohydrodynamics (MHD), heat transfer, mass transport, porous medium resistance, and chemical reaction effects, to simulate realistic blood flow behavior under pathological conditions. The governing equations for momentum, energy, and concentration were formulated using appropriate assumptions for incompressible, electrically conducting blood flow. These equations were non-dimensionalised to identify important controlling parameters such as the Hartmann number (magnetic field strength), Grashof number (thermal buoyancy), solutal Grashof number (concentration buoyancy), Prandtl number, Schmidt number, porosity parameter, and chemical reaction parameter. Analytical methods were employed to obtain solutions, which were further analyzed through graphical and computational techniques. The results reveal that increased sickle cell concentration significantly increases flow resistance, leading to a reduction in velocity and impaired blood circulation, particularly in the presence of arterial narrowing due to atherosclerosis. The temperature gradient plays a dual role: it enhances fluid motion through buoyancy effects while also influencing viscosity and thermal diffusion. The applied magnetic field introduces a Lorentz force that suppresses fluid velocity, thereby providing a potential mechanism for controlling abnormal blood flow. The study demonstrates that the interaction between magnetic field, temperature gradient, and sickle cell concentration has a significant impact on blood flow characteristics in porous, diseased arteries. This work contributes to the advancement of biomedical fluid dynamics by offering a more realistic model for analyzing blood flow in pathological environments. It has potential applications in the design of medical treatments, such as magnetic field-assisted therapy, targeted drug delivery, and improved diagnostic understanding of circulatory disorders associated with sickle cell disease and atherosclerosis.
| Published in | Mathematical Modelling and Applications (Volume 11, Issue 2) |
| DOI | 10.11648/j.mma.20261102.11 |
| Page(s) | 28-40 |
| Creative Commons |
This is an Open Access article, distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium or format, provided the original work is properly cited. |
| Copyright |
Copyright © The Author(s), 2026. Published by Science Publishing Group |
Mathematical Modelling, MHD Blood Flow, Sickle Cell Concentration, Temperature Effect, Porous, Atherosclerotic Channel
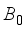 is applied
is applied 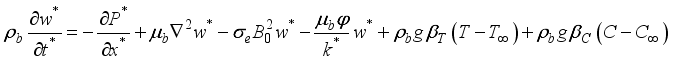 (1)
(1) 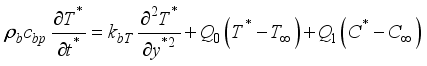 (2)
(2) 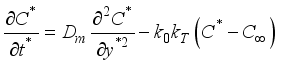 (3)
(3) 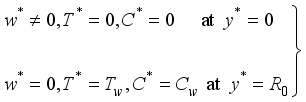 (4)
(4) 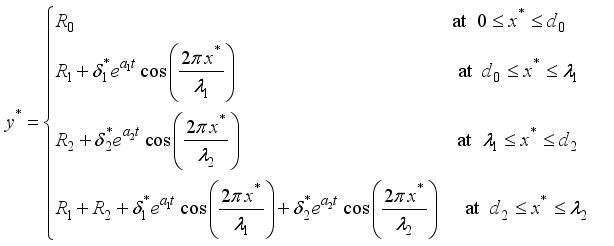 (5)
(5) 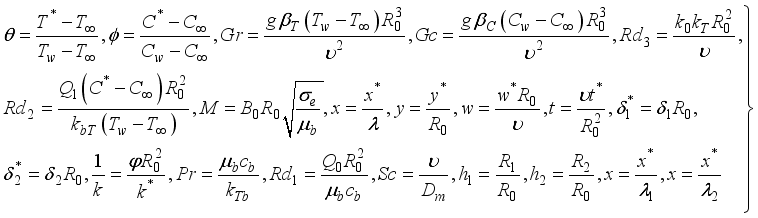 (6)
(6) 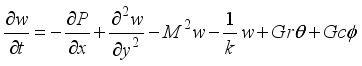 (7)
(7) 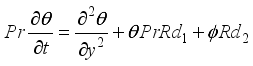 (8)
(8) 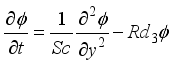 (9)
(9) 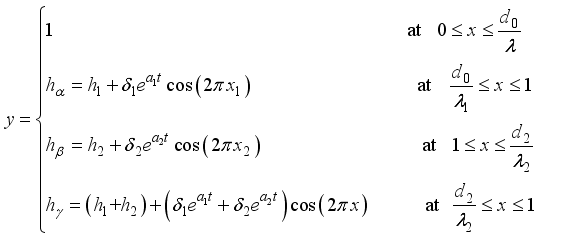 (10)
(10) 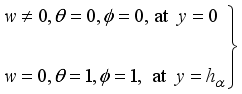 (11)
(11)  (12)
(12)  (13)
(13)  (14)
(14)  (15)
(15) 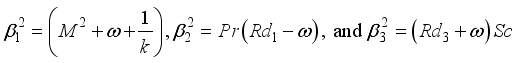
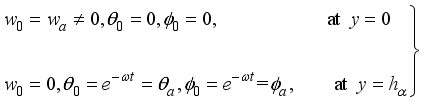 (16)
(16) 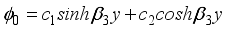 (17)
(17) 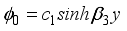 (18)
(18) 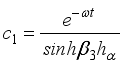
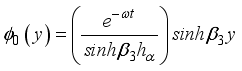 (19)
(19) 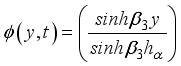 (20)
(20) 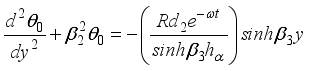 (21)
(21) 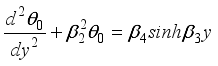 (22)
(22) 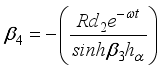
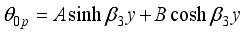 (23)
(23) 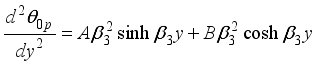 (24)
(24) 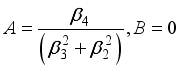 (25)
(25) 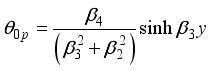 (26)
(26) 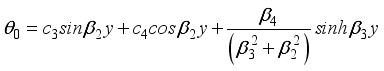 (27)
(27) 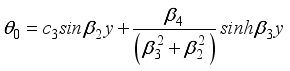 (28)
(28) 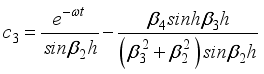 (29)
(29) 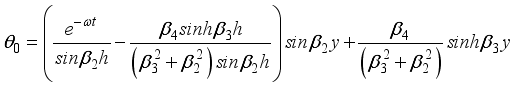 (30)
(30) 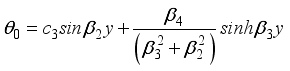 (31)
(31) 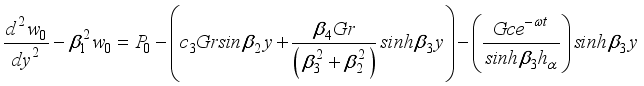 (32)
(32) 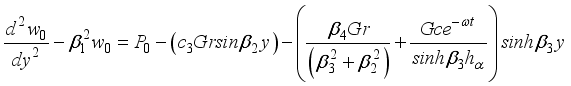 (33)
(33) 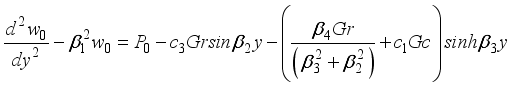 (34)
(34) 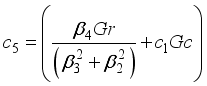 , then equation (34) becomes:
, then equation (34) becomes: 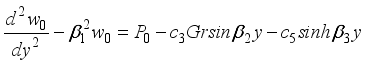 (35)
(35) 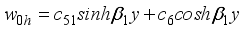 (36)
(36) 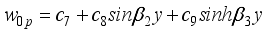 (37)
(37) 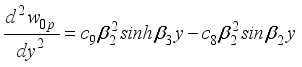 (38)
(38) 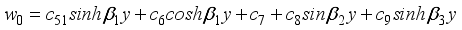 (39)
(39) 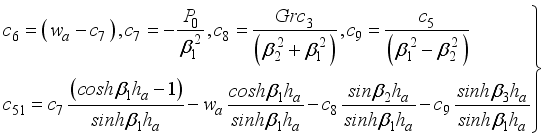 (40)
(40) 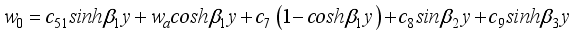 (41)
(41) 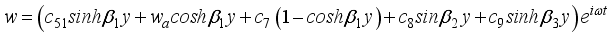 (42)
(42) Dimensional Vertical Distance | |
Dimensional Cell Mass Concentration | |
Dimensional Temperature | |
Far field Cell Mass Concentration | |
Far field Temperature | |
Heat Source Due to Temperature | |
Cell Mass Concentration Source | |
Blood Density | |
Blood Specific Heat Capacity | |
Blood Thermal Conductivity | |
Mass Diffusivity | |
Chemical Reaction | |
Lipid Concentration Treatment | |
Dimensional Time | |
Heat Source Term | |
Cell Mass Concentration Source Term | |
Chemical Reaction Term | |
Dimensionless Cell Mass Concentration | |
Dimensionless Temperature | |
Prandtl Number of Blood | |
Schmidt Number | |
Magnetic Field Parameter | |
Porosity | |
Grashof Number | |
Solutal Grashof Number | |
Oscillatory Frequency Parameter |
| [1] | Abdelsalam, S. I., & Vafai, K. (2017). Particulate suspension effect on peristaltically induced unsteady pulsatile flow in a narrow artery: blood flow model. Mathematical biosciences, 283, 91-105. |
| [2] | Abdullah, I., Amin, N., & Hayat, T. (2011). Magnetohydrodynamic effects on blood flow through an irregular stenosis. Int J Numer Methods Fluids, 67(11), 1624-1636. |
| [3] | Acosta, S., Puelz, C., Rivière, B., Penny, D. J., & Rusin, C. G. (2015). Numerical method of characteristics for one-dimensional blood flow. Journal of computational physics, 294, 96-109. |
| [4] | Ahmed, A., & Nadeem, S. (2016). The study of (Cu, TiO2, Al2O3) nanoparticles as antimicrobials of blood flow through diseased arteries. Journal of Molecular Liquids, 216, 615-623. |
| [5] | Alasakani, K., Tantravahi, S. L. R., & Kumar, P. (2020). An Approach to Identify Significant Parameters in Blood Flow through Human Arteries. Science & Technology Asia, 95-105. |
| [6] | Amos, E., &Ogulu, A. (2003). Magnetic effect on pulsatile flow in a constricted axis-symmetric tube. Indian Journal of Pure & Applied Mathematics, 34(9), 1315-1326. |
| [7] | Bunonyo, K. W., & Amos, E. (2020). Impact of treatment parameter on blood flow in an atherosclerotic artery. American Journal of Theoretical and Applied Statistics, 9(3), 74. |
| [8] | Baroncini, L. A. V., de Castro Sylvestre, L., &Pecoits Filho, R. (2015). Carotid intima-media thickness and carotid plaque represent different adaptive responses to traditional cardiovascular risk factors. International journal of cardiology. Heart &vasculatur8(9), 48-51. |
| [9] | Balkaran, B., Char, G., Morris, J. S., Thomas, P. W., Serjeant, B. E., & Serjeant, G. R. (1992). Stroke in a cohort of patients with homozygous sickle cell disease. The Journal of pediatrics, 120(3), 360-366. |
| [10] | Bunonyo, K. W, Moko, A. B, & Tamuno Boma Odinga (2026) Computational Approach in Mathematical Modeling of Cell Mass Concentration Effect on Heat Transfer Through a Blood Channel. Journal of Mathematical & Computer Applications. SRC/JMCA-281. |
| [11] | Bunonyo, K. W., &Ndu, R. I. (2024). Mathematical Modelling and Numerical Simulation of Haematocrit and Pressure Effects on Blood Flow through Blood Vessel. International Research Journal of Pure and Applied Physics, 11(1), 132-143. |
| [12] | Bunonyo, K. W., & Dagana, J. T. (2024). Mathematical Modelling of the Impact of Mass Concentration on Viscoelastic Fluid Flow through a Non-Porous Channel. Asian Journal of Pure and Applied Mathematics, 6(1), 253-270. Accessed from: |
| [13] | Butter, J. K., Bunonyo, K. W., & Eli, I. C. (2024). Mathematical Modelling of the Thermosolutal Effect on Blood Flow through a Micro-Channel in the Presence of a Magnetic Field. British Journal of Multidisciplinary and Advanced Studies, 5(4), 14-32. |
| [14] | Bunonyo, K. W., Ebiwareme, L., &Iworiso, P. B. (2024). Mathematical modeling of time-dependent concentration of alcohol in the human bloodstream using the eigenvalue method. TWIST, 19(1), 58-64. |
| [15] | Bunonyo, K. W., Ebiwareme, L., &Igodo, A. (2026). Mathematical Examination of MHD Slip Oscillatory Blood Flow Through an Artery in a Magnetic Field. Journal of Physical Mathematics & its Applications. SRC/JPMA-174. (4), 145, 2-6. |
| [16] | Bunonyo, K. W., Peter, B., &Okrinya, A. B. (2026). Application of Piezoelectric Properties in Understanding Voltage Generation, Displacement, and Acceleration via Packed Sickle Cell. Asian Journal of Pure and Applied Mathematics, 8(1), 119-136. |
| [17] | Daniel, M., Szymanik-Grzelak, H., Sierdziński, J., Podsiadły, E., Kowalewska-Młot, M., &Pańczyk-Tomaszewska, M. (2023). Epidemiology and risk factors of UTIs in children—A single-center observation. Journal of personalized medicine, 13(1), 138. |
| [18] | Tziakas, D. N., Chalikias, G., Pavlaki, M., Kareli, D., Gogiraju, R., Hubert, A.,...& Schäfer, K. (2019). Lysed erythrocyte membranes promote vascular calcification: possible role of erythrocyte-derived nitric oxide. Circulation, 139(17), 2032-2048. |
| [19] | Serjeant, G. R. (2005). Mortality from sickle cell disease in Africa. Bmj, 330(7489), 432-433. |
| [20] | Makani, J., Ofori-Acquah, S. F., Nnodu, O., Wonkam, A., & Ohene-Frempong, K. (2013). Sickle cell disease: new opportunities and challenges in Africa. The scientific world journal, 2013(1), 193252. |
| [21] | Rees, D. C., Williams, T. N., & Gladwin, M. T. (2010). Sickle-cell disease. The Lancet, 376(9757), 2018-2031. |
| [22] | Makinde, O. D. (2008). Effects of viscous dissipation and radiation on MHD flow. Applied Mathematics and Computation, 201, 322-329. |
| [23] | Chamkha, A. J. (2003). MHD flow with heat and mass transfer and chemical reaction. International Journal of Engineering Science, 41, 647-662. |
| [24] | Afify, A. A. (2004). Effects of heat and mass transfer on MHD flow. Heat and Mass Transfer, 40, 495-500. |
| [25] | Makinde, O. D., & Aziz, A. (2011). Boundary layer flow with mass transfer. International Journal of Thermal Sciences, 50, 1326-1332. |
| [26] | Raptis, A., Perdikis, C., & Takhar, H. (2004). Effect of thermal radiation on flow. Applied Mathematics and Mechanics, 25, 113-121. |
| [27] | Soundalgekar, V. M. (1979). Oscillatory flow past a plate. Journal of Applied Mechanics. |
| [28] | Nield, D. A., & Bejan, A. (2013). Convection in Porous Media (4th ed.). Springer. |
| [29] | Gebhart, B., Jaluria, Y., Mahajan, R. L., &Sammakia, B. (1988). Buoyancy-Induced Flows and Transport. Hemisphere. |
| [30] | Hartmann, J. (1937). Hg-dynamics I. Kgl. Danske Videnskabernes Selskab. |
| [31] | Shercliff, J. A. (1965). A Textbook of Magnetohydrodynamics. Pergamon. |
| [32] | Ostrach, S. (1953). Natural convection in fluids. NACA Report. |
| [33] | Bejan, A. (2013). Convection Heat Transfer (4th ed.). Wiley. |
| [34] | Shahbaz, H. M., & Ahmad, I. (2024). Numerical treatment for Darcy-Forchheimer flow under MHD effects. Scientific Reports, 14, 31214. |
| [35] | Shahbaz, H. M., & Ahmad, I. (2024). Numerical treatment for Darcy-Forchheimer flow under MHD effects. Scientific Reports, 14, 31214. |
APA Style
Bunonyo, K. W., Moko, A. B., Odinga-Israel, T. B. (2026). Mathematical Modeling of the Impact of Temperature and Elevated Sickle Cell Concentration on MHD Blood Flow Through a Porous Atherosclerotic Channel. Mathematical Modelling and Applications, 11(2), 28-40. https://doi.org/10.11648/j.mma.20261102.11
ACS Style
Bunonyo, K. W.; Moko, A. B.; Odinga-Israel, T. B. Mathematical Modeling of the Impact of Temperature and Elevated Sickle Cell Concentration on MHD Blood Flow Through a Porous Atherosclerotic Channel. Math. Model. Appl. 2026, 11(2), 28-40. doi: 10.11648/j.mma.20261102.11
@article{10.11648/j.mma.20261102.11,
author = {Kubugha Wilcox Bunonyo and Anasuodei Bemoifie Moko and Tamuno Boma Odinga-Israel},
title = {Mathematical Modeling of the Impact of Temperature and Elevated Sickle Cell Concentration on MHD Blood Flow Through a Porous Atherosclerotic Channel},
journal = {Mathematical Modelling and Applications},
volume = {11},
number = {2},
pages = {28-40},
doi = {10.11648/j.mma.20261102.11},
url = {https://doi.org/10.11648/j.mma.20261102.11},
eprint = {https://article.sciencepublishinggroup.com/pdf/10.11648.j.mma.20261102.11},
abstract = {This study offers a comprehensive mathematical and computational investigation of the combined effects of temperature gradient and high sickle cell concentration on blood circulation through a porous atherosclerotic channel in the presence of an applied magnetic field. The model incorporates key physiological and physical mechanisms, including magnetohydrodynamics (MHD), heat transfer, mass transport, porous medium resistance, and chemical reaction effects, to simulate realistic blood flow behavior under pathological conditions. The governing equations for momentum, energy, and concentration were formulated using appropriate assumptions for incompressible, electrically conducting blood flow. These equations were non-dimensionalised to identify important controlling parameters such as the Hartmann number (magnetic field strength), Grashof number (thermal buoyancy), solutal Grashof number (concentration buoyancy), Prandtl number, Schmidt number, porosity parameter, and chemical reaction parameter. Analytical methods were employed to obtain solutions, which were further analyzed through graphical and computational techniques. The results reveal that increased sickle cell concentration significantly increases flow resistance, leading to a reduction in velocity and impaired blood circulation, particularly in the presence of arterial narrowing due to atherosclerosis. The temperature gradient plays a dual role: it enhances fluid motion through buoyancy effects while also influencing viscosity and thermal diffusion. The applied magnetic field introduces a Lorentz force that suppresses fluid velocity, thereby providing a potential mechanism for controlling abnormal blood flow. The study demonstrates that the interaction between magnetic field, temperature gradient, and sickle cell concentration has a significant impact on blood flow characteristics in porous, diseased arteries. This work contributes to the advancement of biomedical fluid dynamics by offering a more realistic model for analyzing blood flow in pathological environments. It has potential applications in the design of medical treatments, such as magnetic field-assisted therapy, targeted drug delivery, and improved diagnostic understanding of circulatory disorders associated with sickle cell disease and atherosclerosis.},
year = {2026}
}
TY - JOUR T1 - Mathematical Modeling of the Impact of Temperature and Elevated Sickle Cell Concentration on MHD Blood Flow Through a Porous Atherosclerotic Channel AU - Kubugha Wilcox Bunonyo AU - Anasuodei Bemoifie Moko AU - Tamuno Boma Odinga-Israel Y1 - 2026/04/10 PY - 2026 N1 - https://doi.org/10.11648/j.mma.20261102.11 DO - 10.11648/j.mma.20261102.11 T2 - Mathematical Modelling and Applications JF - Mathematical Modelling and Applications JO - Mathematical Modelling and Applications SP - 28 EP - 40 PB - Science Publishing Group SN - 2575-1794 UR - https://doi.org/10.11648/j.mma.20261102.11 AB - This study offers a comprehensive mathematical and computational investigation of the combined effects of temperature gradient and high sickle cell concentration on blood circulation through a porous atherosclerotic channel in the presence of an applied magnetic field. The model incorporates key physiological and physical mechanisms, including magnetohydrodynamics (MHD), heat transfer, mass transport, porous medium resistance, and chemical reaction effects, to simulate realistic blood flow behavior under pathological conditions. The governing equations for momentum, energy, and concentration were formulated using appropriate assumptions for incompressible, electrically conducting blood flow. These equations were non-dimensionalised to identify important controlling parameters such as the Hartmann number (magnetic field strength), Grashof number (thermal buoyancy), solutal Grashof number (concentration buoyancy), Prandtl number, Schmidt number, porosity parameter, and chemical reaction parameter. Analytical methods were employed to obtain solutions, which were further analyzed through graphical and computational techniques. The results reveal that increased sickle cell concentration significantly increases flow resistance, leading to a reduction in velocity and impaired blood circulation, particularly in the presence of arterial narrowing due to atherosclerosis. The temperature gradient plays a dual role: it enhances fluid motion through buoyancy effects while also influencing viscosity and thermal diffusion. The applied magnetic field introduces a Lorentz force that suppresses fluid velocity, thereby providing a potential mechanism for controlling abnormal blood flow. The study demonstrates that the interaction between magnetic field, temperature gradient, and sickle cell concentration has a significant impact on blood flow characteristics in porous, diseased arteries. This work contributes to the advancement of biomedical fluid dynamics by offering a more realistic model for analyzing blood flow in pathological environments. It has potential applications in the design of medical treatments, such as magnetic field-assisted therapy, targeted drug delivery, and improved diagnostic understanding of circulatory disorders associated with sickle cell disease and atherosclerosis. VL - 11 IS - 2 ER -