This worked is aimed at studying the thermodynamic and kinetic adsorption of methyl orange (MO) onto activated carbon (AC) obtained from Neem oil cakes (NOC). The ACs were synthesized by chemical activation of Neem oil cakes with H3PO4 of 2, 5 and 10 percent (respectively labeled AC-2, AC-5, and AC-10) followed by pyrolysis at 450°C for 1 hr. Various characterizations of the synthesized ACs include Fourier Transformed Infrared spectroscopy FTIR, microstructural and elemental analyses (SEM/TEM, EDS), pHPZC, moisture content, and iodine and methylene blue adsorption methods were used to determine the surface area. The ACs were employed to adsorb methyl orange (MO) from a synthetic aqueous solution. The results obtained show that: pHPZC was less than 7, indicating that the three activated carbons have predominantly acidic surface. The adsorbents AC-5 and AC-10 have microporous and mesoporous structures respectively, with respective specific surface area by iodine adsorption (SI2) method estimated to be around 688.45 and 689.70 m2/g. The adsorption of MO was pH dependent, with an optimal adsorption at pH =2. The EDS results confirm that these adsorbents are primarily composed of carbon. Results from kinetic studies showed that the adsorption process followed a pseudo second order kinetic model. The experimental data from the equilibrium adsorption of MO on the ACs showed the best fit with the Langmuir isotherm, suggesting monolayer adsorption. Maximum adsorption capacity of 232.558 mg.g-1 was obtained for AC-10. These results show that the adsorption of MO is spontaneous and endothermic. Chemisorption is the predominant mechanism for MO removal on AC-2, AC-5, and AC-10.
| Published in | Science Journal of Chemistry (Volume 14, Issue 2) |
| DOI | 10.11648/j.sjc.20261402.13 |
| Page(s) | 60-74 |
| Creative Commons |
This is an Open Access article, distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium or format, provided the original work is properly cited. |
| Copyright |
Copyright © The Author(s), 2026. Published by Science Publishing Group |
Neem Oil Cake, Activated Carbon, Adsorption, Methyl Orange, Pseudo-second Order, Langmuir Isotherm
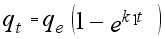 (10)
(10) 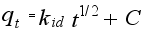 (12)
(12) Activated carbon | AC-2 | AC-5 | AC-10 |
|---|---|---|---|
Phzpc | 4.7 | 5.5 | 5.3 |
Activated carbon | AC-2 | AC-5 | AC-10 |
|---|---|---|---|
Methylene Blue Number (IMB, mg. g-1) | 98.29 | 99.80 | 99.83 |
Specific Surface Area (SMB, m2. g-1) | 60.60 | 61.15 | 62.15 |
Iodine Number (II2, mg. g-1) | 430.45 | 684.50 | 686.24 |
Specific Surface Area (SI2, m2. g-1) | 435.19 | 688.45 | 689.70 |
Activated carbon | AC-2 | AC-5 | AC-10 |
|---|---|---|---|
Moisture Content (%) | 2.7 | 2.5 | 2.3 |
Models | Parameters | AC-2 | AC-5 | AC-10 |
|---|---|---|---|---|
pseudo-first-order | R2 | 0.751 | 0.804 | 0.889 |
K1 () | 0.013 | 0.039 | 0.045 | |
Qe(th) (mg. g-1) | 1.411 | 2.253 | 1.060 | |
Qemax (exp) (mg. g-1) | 29.675 | 29.756 | 29.729 | |
(mg. g-1) | 20.694 | 27.503 | 28.669 | |
t1/2 (min) | 52.900 | 17.960 | 15.400 | |
pseudo-second-order | R2 | 0.999 | 0.999 | 0.999 |
K2 (g. mg-1. min-1) | 0.036 | 0.042 | 0.036 | |
Qe (th) (mg. g-1) | 29.670 | 29.940 | 29.586 | |
Qemax (exp) (mg. g-1) | 29.675 | 29.756 | 29.729 | |
(mg. g-1) | 0.009 | 0.184 | 0.143 | |
h (mg. g-1. min-1) | 31.690 | 37.650 | 31.646 | |
t1/2 (min) | 0.920 | 0.800 | 0.935 | |
intraparticle diffusion | R2 | 0.845 | 0.952 | 0.902 |
Kid ((mg. g-1).) | 0.114 | 0.153 | 0.050 | |
I (mg. g-1) | 28.233 | 28.182 | 28.538 |
Models | Parameters | AC-2 | AC-5 | AC-10 |
|---|---|---|---|---|
Langmuir | 0.993 | 0.998 | 0.997 | |
Qmax(mg/g) | 81.301 | 207.270 | 232.558 | |
KL (L. | 0.062 | 0.061 | 0.266 | |
RL | 0.151 | 0.154 | 0.040 | |
Freundlich | 0.965 | 0.995 | 0.976 | |
Kf (..) | 13.121 | 16.458 | 17.782 | |
Nf | 1.037 | 1.313 | 1.583 | |
1/nf | 0.964 | 0.762 | 0.632 |
AC | Temperature (K) | G (kJ.mol-1) | H (kJ.mol-1) | S (kJ.mol-1) |
|---|---|---|---|---|
AC-2 | 308.150 | -8.010 | 0.253 | 0.027 |
318.150 | -8.278 | |||
328.150 | -8.546 | |||
338.150 | -8.814 | |||
348.50 | -9.082 | |||
AC-5 | 308.150 | -12.191 | 0.744 | 0.042 |
318.150 | -12.612 | |||
328.150 | -13.031 | |||
338.150 | -13.451 | |||
348.150 | -13.871 | |||
AC-10 | 308.150 | -11.765 | 0.680 | 0.040 |
318.150 | -12.169 | |||
328.150 | -12.573 | |||
338.150 | -12.976 | |||
348.150 | -13.380 |
AC | Activated Carbon |
NOC | Neem Oil Cakes |
MO | Methyl Orange |
MB | Methylene Blue |
FTIR | Fourier Transform Infrared |
SEM/EDS | Scanning Electronic Microscopy Coupled Energy Dispersive X Ray |
∆ G | Free Enthalpy Variation |
∆ H | Enthalpy Variation |
∆S | Entropy Variation |
| [1] | Hayat M., Shah A., Nisar J., Shah I., Haleem A., Ashiq M.N., “A novel electrochemical sensing platform for the sensitive detection and degradation monitoring of methylene blue“. Catalysts, vol. 12, pp. 306–319, 2022. |
| [2] | Garg V.K., Amita M., Kumar R., Gupta R., “Basic dye (methyl ene blue) removal from simulated wastewater by adsorption using Indian Rosewood sawdust: a timber industry waste“. Dyes Pigm, vol. 63, pp. 243–250, |
| [3] | Veisi H., Razeghi S., Mohammadi P., Hemmati S., “Silver nanoparticles decorated on thiol-modified magnetite nanoparticles (Fe3O4/SiO2-Pr-S-Ag) as a recyclable nanocatalyst for degradation of organic dyes“. Mater Sci Eng C Mater Biol Appl, vol. 97, pp. 624–631, |
| [4] | Pacheco-Álvarez M.O., Picos A., Pérez-Segura T., Peralta-Hernán dez J.M., “Proposal for highly efficient electrochemical dis coloration and degradation of azo dyes with parallel arrangement electrodes“. J Electroanal Chem, vol. 838, pp. 195–203, |
| [5] | Du J., Bao J., Lu C., Werner D., “Reductive sequestration of chromate by hierarchical FeS@Fe0 particles“. Water Res, vol. 102, pp. 73–81. |
| [6] | Pathania D., Bhat V.S., Mannekote Shivanna J., Sriram G., Kurkuri M., Hegd G.,“Garlic peel based mesoporous carbon nano spheres for an effective removal of malachite green dye from aqueous solutions: detailed isotherms and kinetics”. Spectrochi Acta -A: Mol Biomol Spectrosc, vol. 276, pp. 121197, |
| [7] | Samsami S., Mohamadizaniani M., Sarrafzadeh M.H., Rene E.R., Firoozbahr M.,“ Recent advances in the treatment of dye containing wastewater from textile industries: overview and per spectives“. Process Saf Environ Prot, vol. 143, pp. 138–163, |
| [8] | Vendemiatti J.A.S., Camparotto N.G., Vidal C., Cristale J., Agapito E.V.M., Oliveira Á.C., Rodrigues E.A., Montagner C.C., Umbuzeiro G.A., Prediger P., “ New benzotriazoles generated during textile dyeing process: synthesis, hazard, water occurrence and aquatic risk assessment“. J Hazard Mater vol. 403, pp. 123732, |
| [9] | Naziha L., Smail T., Hadi D., “High efficiency adsorption performance of basic dye (crystal violet) onto algerian montmorillonite“. Chem. Eng. Biotechnol. Food Ind, vol. 25, pp. 001– 018, 2024. |
| [10] | Eman M., Abd El-Monaem H.M., Elshishini S.S., Bakr H.G., ElAqapa M.H., Gangadhar A., Gehan M., El-Subruiti A.M., Omer,Abdelazeem S., Eltaweil. “A comprehensive review on LDH-based catalysts to activate persulfates for the degradation of organic pollutants, NPJ Clean“. Water. vol. 6, pp. 34, |
| [11] | Razali N. S., Abdulhameed A. S., Jawad A. H., Alothman Z. A., Yousef T. A., Al Duaij O. K., Alsaiari N. S., “ High-Surface-Area-Activated Carbon Derived from Mango Peels and Seeds Wastes via Microwave-Induced ZnCl2 Activation fo Adsorption of Methylene Blue Dye Molecules: Statistical Optimization and Mechanism“. Mol., vol. 27, pp. 6947. |
| [12] | Mahmoodi N.M., Karimi B., Mazarji M., Moghtaderi H., “ Cadmium selenide quantum dot-zinc oxide composite: synthesis, characterization, dye removal ability with UV irradiation, and antibacterial activity as a safe and high-performance photocatalyst“. J Photochem Photobiol B Biol, vol. 188, pp. 19–27, |
| [13] | Rabeie B., Mahmoodi N.M., “ Heterogeneous MIL-88A on MIL-88B hybrid: a promising eco-friendly hybrid from green syn thesis to dual application (adsorption and photocatalysis) in tetra cycline and dyes removal“. J Colloid Interface Sci, vol. 654, pp. 495–522, |
| [14] | Dutta S., Gupta B., Srivastava S.K., Gupta A.K., “ Recent advances on the removal of dyes from wastewater using various adsorbents: a critical review“. Mater Adv, vol. 2 pp. 4497–4531, |
| [15] | Patawat C., Silakate K., Chuan Udom S., Supanchaiyamat N., Hunt A.J., Ngernyen Y., “ Preparation of activated carbon from Diptero carpus alatus fruit and its application for methylene blue adsorption“. RSC Adv, vol. 10 pp. 21082–21091, |
| [16] | Mahmoodi N.M., Maghsoodi A., “ Kinetics and isotherm of cationic dye removal from multicomponent system using the syn thesized silica nanoparticle“. Desalination Water Treat, vol. 54 pp. 562–571, |
| [17] | Mahmoodi N.M., Mokhtari-Shourijeh Z., (2016) “Modified poly (vinyl alcohol)-triethylenetetramine nanofiber by glutaraldehyde: preparation and dye removal ability from wastewater“. Desalination Water Treat, vol. 57 pp. 20076–20083, |
| [18] | Katheresan V., Kansedo J., Lau S.Y., “Efficiency of various recent wastewater dye removal methods: a review“. J Environ Chem Eng, vol. 6 pp. 4676–4697, |
| [19] | Saepurahman., Singaravel G.P., Hashaikeh R., “Fabrication of electrospun LTL zeolite fibers and their application for dye removal“. J. Mater. Sci, vol. 51, pp. 1133–1141. |
| [20] | Liu Z., Khan T. A., Islam M. D. A., Tabrez U., “A review on the treatment of dyes inprinting and dyeing wastewater by plant biomass carbon“. Bio. Tech, vol. 354, pp. 127168, |
| [21] | Tabrez A.K.., Rumana R., Equbal A.K., “ Decolorization of bismarck brown R and crystal violet in liquid phase using modified pea peels: non-linear isotherm and kinetics modeling“, Model. Earth Syst. Environ, vol. 2, pp. 1-11. |
| [22] | Sakin O.O., Hussein M.A., Hussein B. H. M., Mgaidi A., “Adsorption thermodynamics of cationic dyes (methylene blue and crystal violet) to a natural clay mineral from aqueous solution“. Arab. J. Chem, vol. 11, pp. 615-623. |
| [23] | Indujalekshmi J., Arsha M.S., Biju V., “KOH-mediated structural modification of activated charcoal by heat treatment for the efficient adsorption of organic dyes“. Appl. Surf. Sci. Adv, vol. 19, pp. 100566. |
| [24] | Tiotsop I.H.K., Donald R.T.T., Doungmo G., et al., “ Preparation and characterization of activated carbons from bitter kola (Garcinia kola) nut shells by chemical activation method using H3PO4; KOH and ZnCl2“. Chemical Sci. Inter. J, vol. 23, pp. 1-15, |
| [25] | Sun S., Zhu Y., Gu Z., Chu H., Hu C., Gao L., Zhao X., “Adsorption of crystal violet on activated bamboo fiber powder from water: preparation, characterization, kinetics and isotherms“. Rsc. Adv, vol. 13, pp. 6108–6123. |
| [26] | Sadoq M., Atlas H., Imame S., Kali A., Amar A., Loulidi I., Jabri M., Sadoq B. E., Ouchabi M., Abdullah P. S., Boukhlifi F., “Elimination of crystal violet from aqueous solution by adsorption on naturel polysaccharide: Kinetic, isotherm, thermodynamic studie and mechanism analysis“. Arab. J. Chem, vol. 17, pp. 105453. |
| [27] | Hosseinabadi-Farahani Z., Mahmoodi N.M., Hosseini-Monfared H., “Preparation of surface functionalized graphene oxide nanosheet and its multicomponent dye removal ability from waste water“. Fibers Polymers, vol. 16, pp. 1035–1047, |
| [28] | Hosseinabadi-Farahani Z, Hosseini-Monfared H., Mahmoodi N.M.,“ Graphene oxide nanosheet: preparation and dye removal from binary system colored wastewater“. Desalination Water Treat, vol. 56, pp. 2382–2394, |
| [29] | Mokhtari-Shourijeh Z., SepidehLangari L.M., Mahmoodi N.M.,“Synthesis of porous aminated PAN/PVDF composite nanofibers by electrospinning: characterization and direct red 23 removal“. J Environ Chem Eng, vol. 8, pp. 103876, |
| [30] |
Mahmoodi N.M., Hosseinabadi-Farahani Z., Chamani H., Dye adsorption from single and binary systems using NiO-MnO2 nanocomposite and artificial neural network modeling“. Environ Prog Sustain, vol. 36 pp. 111–119,
https://doi.org/10.1002/ep.12452 , 2017. |
| [31] | Ho Y.S., McKay.,G “Sorption of dye from aqueous solution by peat“. Chem Eng J,vol. 70 pp. 115–124, |
| [32] | Aravind Kumar J., Krithiga T., Narendrakumar G., Prakash P., Bala sankar K., Sathish S., Prabu D., Purna Pushkala D., Marraiki N., Ramu A.G., Choi D., “ Effect of Ca2+ ions on naphthalene adsorption/desorption onto calcium oxide nanoparticle: adsorption isotherm, kinetics and regeneration studies“. Environ Res, vol. 204 pp. 112070, |
| [33] | Ho Y., “Review of second-order models for adsorption systems“. J. Hazard. Mater, vol. 136, pp. 681–689, |
| [34] | Mazaheri H., Ghaedi M., Hajati S., Dashtian K., Purkait M.K., “Simultaneous removal of methylene blue and Pb2+ ions using ruthenium nanoparticle-loaded activated carbon: response surface methodology“. RSC. Adv vol. 5, pp. 83427–83435, |
| [35] | Luttah I., Onunga D.O., Shikuku V.O., Otieno B., Kowenje C.O., “Removal of endosulfan from water by municipal waste incineration flyash-based geopolymers: Adsorption kinetics, isotherms, and thermodynamics“. Front. Environ. Chem, vol. 4. |
| [36] | Abdulkareem A.S., Hamzat W.A., Tijani J.O., Egbosiuba T.C., Mustapha S., Abubakre O.K., Okafor B.O., Babayemi A.K., “Isotherm, kinetics, thermodynamics and mechanism of metal ions adsorption from electroplating wastewater using treated and functional ized carbon nanotubes“. J Environ Chem Eng, vol. 11, pp. 109180, |
| [37] | Mbachu C.A., Babayemi A.K., Egbosiuba T.C., Ike J.I., Ani I.J., Mustapha S., “Green synthesis of iron oxide nanoparticles by Tagu chi design of experiment method for effective adsorption of methylene blue and methyl orange from textile wastewater“. Res Eng Des, vol. 19 pp. 101198. |
| [38] | Ndongo G.K., Ndi J.N., Ketcha J.M., “Ferromagnetic Activated Carbon from Cassava (Manihot dulcis) Peels Activated by Iron (III) Chloride: Synthesis and Characterization“. Bio. Res. vol. 15, pp. 2133-2146, 2020. |
| [39] | Mbouombouo B.J., Ngassa P.G., Mabou L.J., Zapenaha P.H., Maffeu E.J., Mondahchouo S., Doungmo G., Victor O.S., Tchieta G.P., Kamdem W.F.,. “Removal of crystal violet by TiO2 loaded alkali ‑ activated carbon hybrid material from Raphia farinifera fruit kernels: surface chemistry, parameters and mechanisms“. Biomass. Conv. Bioref., pp. 1-22. |
| [40] | Haleema M., C., Iqra R. H., Adeel A., Ahmed, Al-Shoaibi, Srinivasakannan C., “Enhanced moisture adsorption of activated carbon through surface modification“. Results Surf. Interfaces, vol. 14, pp. 100170, |
| [41] | Katarzyna J., Barbara C., “Adsorption properties of biochars obtained by KOH activation“.. Adsorption, vol. 30, pp. 167–183, |
| [42] | Abdullah C.A., Ömer Ş., Orhan B., et al., “Surface and porous characterization of activated carbon prepared from pyrolysis of biomass by two-stage procedure at low activation temperature and it's the adsorption of iodine“. Journal of Analytical and Applied Pyrolysis, vol. 104, pp. 378-383, |
| [43] | Chao D., Bo L., Jian H., et al., “ b c,dwnvbn,; n,n,; ,n n, n,,nn, Determination of iodine number of activated carbon by the method of ultraviolet–visible spectroscopy“. Materials Letters, vol. 285, pp. 129137, |
| [44] | Ndi J.S., “Textural properties and adsorption characteristics of activated carbon prepared from cola (C. Acuminata) nut shells: Application for the elimination of methylene blue from aqueous solution". Thèse Doctorat Univ. Yaoundé I, Cameroun, pp. 200, 2014. |
| [45] | Ouéda N, Igor W.O., Yvonne L.B., “Effets des paramètres physico-chimiques de charbons actifs de balle de riz sur la capacité d’adsorption du glycérol“, J. Soc. Ouest-Afr. Chim. vol. 047 pp. 61-72, 2019. |
| [46] | To M., Sze C., Lin K., Mckay G., “Mechanistic study of atenolol, acebutolol and carbamazepine adsorption on waste biomass derived activated carbon“. J. Mol. Liq, vol. 241, pp. 386–398, |
| [47] | Essekria A., Hsinia A., Naciri Y., Laabd M., Ajmal Z., El Ouardi M., Ait Addi A., Albourine A., “Novel citric acid-functionalized brown algae with a high removal efficiency of crystal violet dye from colored wastewaters: insights into equilibrium, adsorption mechanism, and reusability“. Int. J. Phytoremed, |
| [48] | Batool M., Javed T., Wasim M., Zafar S., “Exploring the usability of Cedrus deodara sawdust for decontamination of wastewater containing crystal violet dye. Desalin“. Water Treat, vol. 224, pp. 433–448, |
APA Style
Mabou, J. B. L., Nforna, E. A., Makota, S., Poumve, H. Z., Malama, S., et al. (2026). A Study on the Adsorption of Methyl Orange in Aqueous Solution by Activated Carbon Prepared from Neem Oil Cakes: Kinetic and Thermodynamic Analyses. Science Journal of Chemistry, 14(2), 60-74. https://doi.org/10.11648/j.sjc.20261402.13
ACS Style
Mabou, J. B. L.; Nforna, E. A.; Makota, S.; Poumve, H. Z.; Malama, S., et al. A Study on the Adsorption of Methyl Orange in Aqueous Solution by Activated Carbon Prepared from Neem Oil Cakes: Kinetic and Thermodynamic Analyses. Sci. J. Chem. 2026, 14(2), 60-74. doi: 10.11648/j.sjc.20261402.13
@article{10.11648/j.sjc.20261402.13,
author = {Jules Blaise Leuna Mabou and Edwin Akongnwi Nforna and Suzanne Makota and Harlette Zapenaha Poumve and Simon Malama and Jacques Bomiko Mbouombouo and Lincold Nintedem Magapgie and Pierre Gerard Tchieta},
title = {A Study on the Adsorption of Methyl Orange in Aqueous Solution by Activated Carbon Prepared from Neem Oil Cakes: Kinetic and Thermodynamic Analyses},
journal = {Science Journal of Chemistry},
volume = {14},
number = {2},
pages = {60-74},
doi = {10.11648/j.sjc.20261402.13},
url = {https://doi.org/10.11648/j.sjc.20261402.13},
eprint = {https://article.sciencepublishinggroup.com/pdf/10.11648.j.sjc.20261402.13},
abstract = {This worked is aimed at studying the thermodynamic and kinetic adsorption of methyl orange (MO) onto activated carbon (AC) obtained from Neem oil cakes (NOC). The ACs were synthesized by chemical activation of Neem oil cakes with H3PO4 of 2, 5 and 10 percent (respectively labeled AC-2, AC-5, and AC-10) followed by pyrolysis at 450°C for 1 hr. Various characterizations of the synthesized ACs include Fourier Transformed Infrared spectroscopy FTIR, microstructural and elemental analyses (SEM/TEM, EDS), pHPZC, moisture content, and iodine and methylene blue adsorption methods were used to determine the surface area. The ACs were employed to adsorb methyl orange (MO) from a synthetic aqueous solution. The results obtained show that: pHPZC was less than 7, indicating that the three activated carbons have predominantly acidic surface. The adsorbents AC-5 and AC-10 have microporous and mesoporous structures respectively, with respective specific surface area by iodine adsorption (SI2) method estimated to be around 688.45 and 689.70 m2/g. The adsorption of MO was pH dependent, with an optimal adsorption at pH =2. The EDS results confirm that these adsorbents are primarily composed of carbon. Results from kinetic studies showed that the adsorption process followed a pseudo second order kinetic model. The experimental data from the equilibrium adsorption of MO on the ACs showed the best fit with the Langmuir isotherm, suggesting monolayer adsorption. Maximum adsorption capacity of 232.558 mg.g-1 was obtained for AC-10. These results show that the adsorption of MO is spontaneous and endothermic. Chemisorption is the predominant mechanism for MO removal on AC-2, AC-5, and AC-10.},
year = {2026}
}
TY - JOUR T1 - A Study on the Adsorption of Methyl Orange in Aqueous Solution by Activated Carbon Prepared from Neem Oil Cakes: Kinetic and Thermodynamic Analyses AU - Jules Blaise Leuna Mabou AU - Edwin Akongnwi Nforna AU - Suzanne Makota AU - Harlette Zapenaha Poumve AU - Simon Malama AU - Jacques Bomiko Mbouombouo AU - Lincold Nintedem Magapgie AU - Pierre Gerard Tchieta Y1 - 2026/04/30 PY - 2026 N1 - https://doi.org/10.11648/j.sjc.20261402.13 DO - 10.11648/j.sjc.20261402.13 T2 - Science Journal of Chemistry JF - Science Journal of Chemistry JO - Science Journal of Chemistry SP - 60 EP - 74 PB - Science Publishing Group SN - 2330-099X UR - https://doi.org/10.11648/j.sjc.20261402.13 AB - This worked is aimed at studying the thermodynamic and kinetic adsorption of methyl orange (MO) onto activated carbon (AC) obtained from Neem oil cakes (NOC). The ACs were synthesized by chemical activation of Neem oil cakes with H3PO4 of 2, 5 and 10 percent (respectively labeled AC-2, AC-5, and AC-10) followed by pyrolysis at 450°C for 1 hr. Various characterizations of the synthesized ACs include Fourier Transformed Infrared spectroscopy FTIR, microstructural and elemental analyses (SEM/TEM, EDS), pHPZC, moisture content, and iodine and methylene blue adsorption methods were used to determine the surface area. The ACs were employed to adsorb methyl orange (MO) from a synthetic aqueous solution. The results obtained show that: pHPZC was less than 7, indicating that the three activated carbons have predominantly acidic surface. The adsorbents AC-5 and AC-10 have microporous and mesoporous structures respectively, with respective specific surface area by iodine adsorption (SI2) method estimated to be around 688.45 and 689.70 m2/g. The adsorption of MO was pH dependent, with an optimal adsorption at pH =2. The EDS results confirm that these adsorbents are primarily composed of carbon. Results from kinetic studies showed that the adsorption process followed a pseudo second order kinetic model. The experimental data from the equilibrium adsorption of MO on the ACs showed the best fit with the Langmuir isotherm, suggesting monolayer adsorption. Maximum adsorption capacity of 232.558 mg.g-1 was obtained for AC-10. These results show that the adsorption of MO is spontaneous and endothermic. Chemisorption is the predominant mechanism for MO removal on AC-2, AC-5, and AC-10. VL - 14 IS - 2 ER -